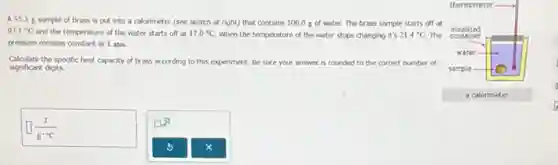
A 55.3 g sample of brass is put into a calorimeter (see sketch at right) that contains 100.0 g of water. The brass sample starts off at 97.1^circ C and the temperature of the water starts off at 17.0^circ C When the temperature of the water stops changing it's 21.4^circ C The pressure remains constant at 1 atm. Calculate the specific heat capacity of brass according to this experiment. Be sure your answer is rounded to the correct number of significant digits.
Solution4.3(344 votes)
Answer
Explanation
Similar Questions
15) (3pts) Hydrolysis leads to formation of (mark all that are correct) A) monomers B) polymers D) water
When two substances are not chemically bonded but are mixed together, the mixture is called an emulsion. TRUE FALSE
A common use of halite is in drywall and plaster. Select one: True False
Find the error with subject-verb agreement.Select the Incorrect verb and type it correctly. The number of protons in an atom are expressed by that element's atomic number on the periodic table.
5. SEP Make Observations Discuss what happened after the sodium alginate solution was added to the calcium chloride solution? __
During brazing, which of the following may present health hazards? Zinc Beryllium Cadmium or phosphorus All are correct
6. What is the polarity of the water molecule compared to? tug of war between two football players tug of war between a football player and toddler tug of war between two toddlers
Water has a low specific heat capacity. True False
Which choice represents a group of elements with common characteristics or properties? silver, iodine, helium oxygen, hydrogen, lithium copper, oxygen, silver copper, gold, silver
Which particles determine the affinity of atoms to each other and thus the likelihood of a chemical reaction? Protons Neutrons Electrons Nucleus









