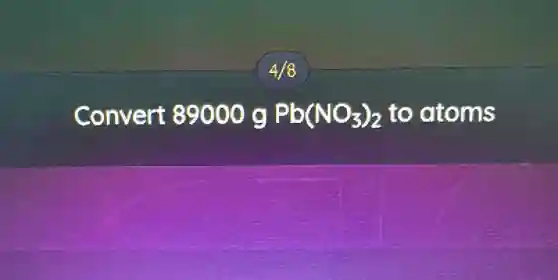
Convert 89000 g Pb(NO_(3))_(2) to atoms
Solution4.0(274 votes)
Answer
Explanation
Similar Questions
Given the following reactions and corresponding $\Delta H$ values, find the $\Delta H$ (to the nearest whole \#) for the reaction: $H_{2}SO_{4}(l)\rightarrow SO_{3}(g)+H_{2}O(g)$ $H_{2}S(g)+2O_{2}(g)\rightarrow H_{2}SO_{4}(l)$ $\Delta H=-235kJ$ $H_{2}S(g)+2O_{2}(g)\rightarrow SO_{3}(g)+H_{2}O(l)$ $\Delta H=-207kJ$ $H_{2}O(l)\rightarrow H_{2}O(g)$ $\Delta H=44kJ$ $\square $
Convert $4.68\times 10^{25}$ atoms $PBr_{3}$ to mol
Which of the following best describes a mixture? a material that is made up of many molecules a substance that can be changed into another substance a material that contains two or more substances a substance that has been broken down into Its smallest parts
What is used to of a hope of sixes in a a compound heard on the chermal reme? the periodic table prefixes chemical symbols the IUP AC
40. Using the hyphen form of symbol writing write the name of this atom. ${}_{9}^{19}F^{-1}$
Which of the following will not increase 1 point the rate of a chemical reaction? increasing surface area stirring decreasing the temperature increasing the concentration A __ is a substance that increases the rate of a reaction without being consumed. __ 1 point A(n) __ slows down the rate of a reaction. __ is a substance that 1 point
Use molecular orbital theory to determine the bond order in $H_{2}$ o 2 1 3
What is enthalpy? the heat absorbed or released during a phase change the heat content.of a system at constant pressure the heat absorbed during an endothermic reaction the heat given off during an exothermic reaction
1. What are two ways in which real gases differ from ideal gases? $\square $
What partie should be used for the caygin akan when maning the characal formuls $SO_{2}?$ $\square $ disappointed mono- pent.









