Which example is a weak base? NaOH KOH CaO CO_(3)^2-
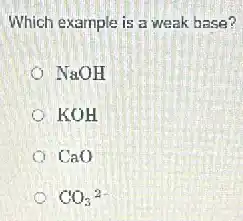
Solution4.6(129 votes)
Answer
Explanation
Similar Questions
$3NH_{4}NO_{3}+Na_{3}PO_{4}\rightarrow (NH_{4})_{3}PO_{4}+3NaNO_{3}$ 1. You have 30 grams of ammonium nitrate and 50.0 grams of sodium phosphate, What is the limiting reactant? 2. What is the excess reactant? 3. What is the maximum amount of each product produced?
6. How much $2\% $ acetic acid solution can be made from 75 mL of a $100\% $ stock solution? 7. How much $30\% $ stock sodium hydroxide solution should be added to 500 mL of water to make a $5\% $ sodium hydroxide solution?
Which property does an acid have? feels slippery tastes bitter D tastes salty reacts with metal
Which salts will he more soluble in an acidic solution than in pure water? CuCN $Zn(OH)_{2}$ $CaSO_{4}$ AgBr $KClO_{4}$
Which example is an endothermic process? burning wood in a campfire a liquid evaporating into a gas strong hydrochloric acid dissolving in water a piece of iron rusting
Spectator ions are included in net ionic equations. a TRUE b FALSE
2. What is the gram-molecular mass of the compound with the formula $CH_{3}COOH$ 22.49 44.09 48.09 60.09
3. What is the number of moles of $N_{2}(g)$ that contains $9.03\times 10^{23}$ molecules? 0.667 1.50 33.6 42.0
Calculate how many atoms of each element are present in the following compounds. 1. $NaHCO_{3}$ 2. $C_{2}H_{4}O_{2}$ 3. $Mg(OH)_{2}$ 4. $3H_{3}PO_{4}$ 5. $2H_{2}SO_{4}$
$HCO_{3}+Mg(OH)_{2}\rightarrow H_{2}O+MgCO_{3}$









